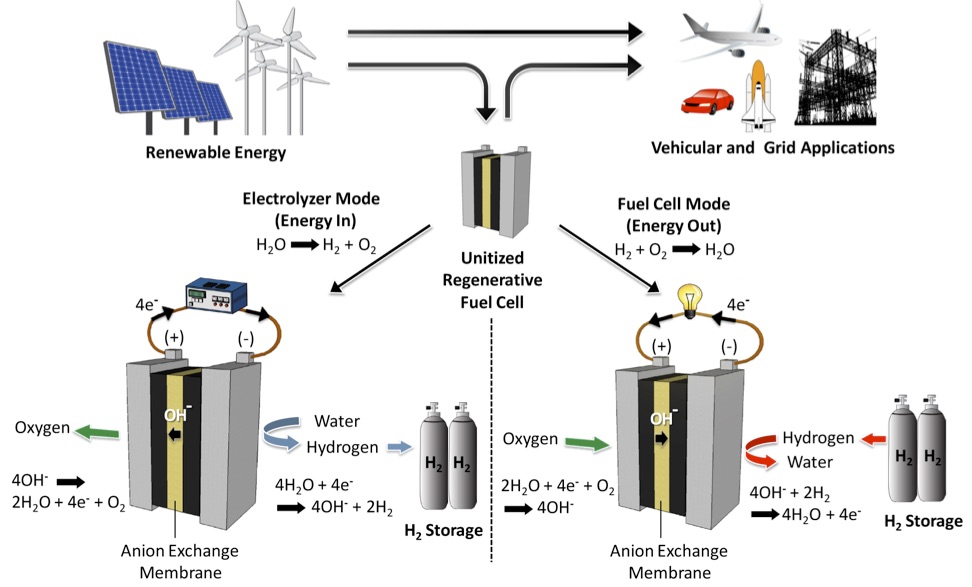
Above: Schematic representation of an AEM-URFC as an energy storage
device for vehicular and grid applications. The AEM-URFC stores renewable
energy as H2 while in electrolyzer mode and then uses that H2 to produce
electric energy on-demand when in fuel cell mode.
Our device development efforts involve incorporating electrocatalysts
into energy storage and conversion systems. For example, we have developed a
precious-metal-free, low-temperature unitized regenerative fuel cell based
on H2O-H2-O2 interconversion chemistries that could potentially address the
intermittency issue afflicting renewable energy as shown in the figure above [1]. We
have also engineered polymer electrolyte membrane electrolyzers that uses
cost-effective molybdenum sulfides as alternative to precious-metal Pt to
drive the hydrogen evolution reaction at the cathode [2]. Future work includes
developing devices that couple either CO2 reduction or N2 reduction with water
oxidation to generate liquid fuels or ammonia feedstock respectively.
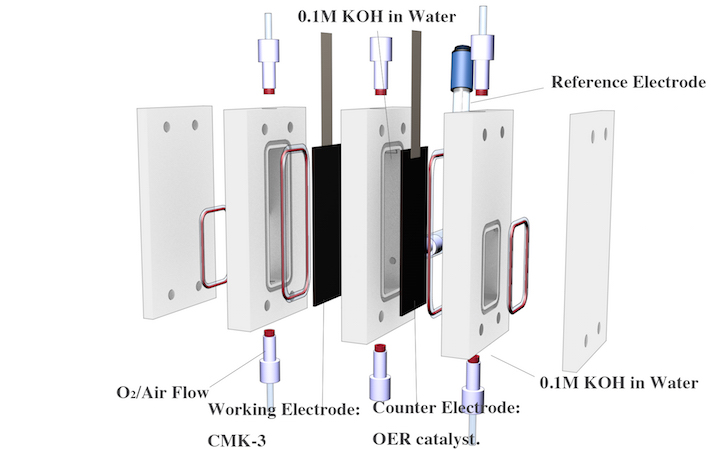
In addition, we are developing devices for hydrogen peroxide production. The development of small-scale, decentralized reactors for H2O2 production that can be coupled to renewable energy sources are of particular interest for water purification in the developing world and other potential decentralization application. We are currently investigating carbon-based catalysts that demonstrate excellent performance for H2O2 production in alkaline to neutral conditions. We have also designed, low-cost, membrane-free reactor design represents a step towards a continuous, modular-scale, de-centralized production of H2O2.
[1] J.W.D. Ng, Y. Gorlin, T. Hatsukade, and T.F. Jaramillo*, “A precious-metal-free regenerative fuel cell for storing renewable electricity”, Advanced Energy Materials, 3 (12), 1545-1550 (2013).
[2] J.W.D. Ng, T.R. Hellstern, J. Kibsgaard, A.C. Hinckley, J.D. Benck, T.F. Jaramillo, “Polymer Electrolyte Membrane Electrolyzers Utilizing Non-precious Mo-based Hydrogen Evolution Catalysts”, Accepted in ChemSusChem (2015).